REVIEW PAPER
New antibiotics in clinical trials – prospects for antibacterial treatment development
1
Warszawski Uniwersytet Medyczny, Polska
2
Katedra i Zakład Farmakologii Doświadczalnej i Klinicznej, Warszawski Uniwersytet Medyczny, Polska
Corresponding author
Adriana Wawer
Katedra i Zakład Farmakologii Doświadczalnej i Klinicznej, Warszawski Uniwersytet Medyczny, Polska
Katedra i Zakład Farmakologii Doświadczalnej i Klinicznej, Warszawski Uniwersytet Medyczny, Polska
Med Og Nauk Zdr. 2023;29(2):73-78
KEYWORDS
TOPICS
ABSTRACT
Introduction and objective:
The discovery of new compounds with antibacterial properties is one of the most important achievements of modern medicine. Unfortunately, the development of new resistance mechanisms and the relatively easy transfer of resistance genes between bacterial cells is now a huge threat and makes many antibiotics ineffective. There is a need to search for new chemical compounds with antibacterial activity, especially against pathogens with critical priority (multidrug-resistant bacteria – Acinetobacter baumanii and Pseudomonas aeruginosa resistant to carbapenems and Enterobacteriaceae producing β-lactamases and resistant to carbapenems and third generation cephalosporins). The aim of this article is to review current clinical trials on new antibacterial compounds.
Review methods:
A literature review was conducted using the PubMed, Google Scholar, Medline databases and the clinicaltrials.gov database, which collects data on clinical trials.
Abbreviated description of the state of knowledge:
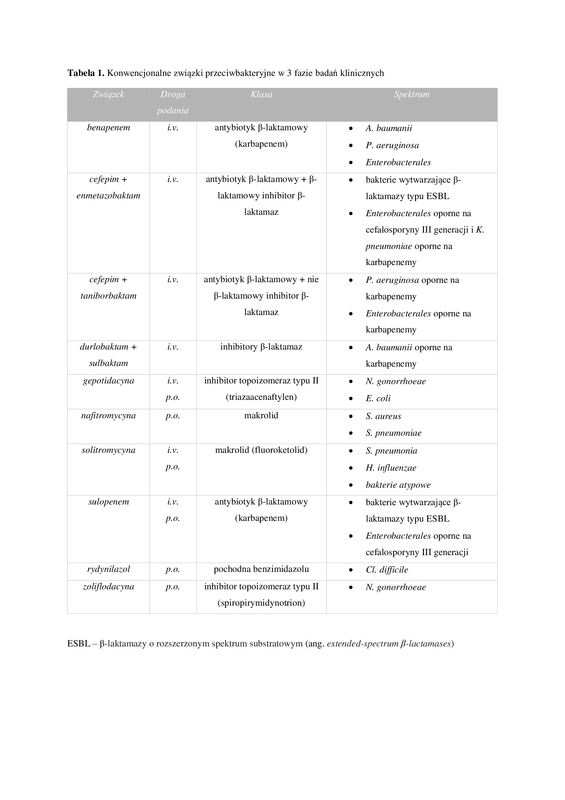
According to a report published by the World Health Organization (WHO), 46 antibiotics are in various stages of clinical trials, of which 28 are potentially active against pathogens on the priority list. However, only 17 of these antibiotics fulfil at least one innovation criteria.
Summary:
The growing prevalence of multidrug-resistant bacteria is now an urgent public health issue. Antibiotic resistance of bacteria is a challenge that modern medicine has to face, it is increasingly said that we are approaching the beginning of the post-antibiotic era. According to the World Health Organization, recently approved antibiotics and those currently in clinical trials are not sufficient to spread drug resistance.
The discovery of new compounds with antibacterial properties is one of the most important achievements of modern medicine. Unfortunately, the development of new resistance mechanisms and the relatively easy transfer of resistance genes between bacterial cells is now a huge threat and makes many antibiotics ineffective. There is a need to search for new chemical compounds with antibacterial activity, especially against pathogens with critical priority (multidrug-resistant bacteria – Acinetobacter baumanii and Pseudomonas aeruginosa resistant to carbapenems and Enterobacteriaceae producing β-lactamases and resistant to carbapenems and third generation cephalosporins). The aim of this article is to review current clinical trials on new antibacterial compounds.
Review methods:
A literature review was conducted using the PubMed, Google Scholar, Medline databases and the clinicaltrials.gov database, which collects data on clinical trials.
Abbreviated description of the state of knowledge:
According to a report published by the World Health Organization (WHO), 46 antibiotics are in various stages of clinical trials, of which 28 are potentially active against pathogens on the priority list. However, only 17 of these antibiotics fulfil at least one innovation criteria.
Summary:
The growing prevalence of multidrug-resistant bacteria is now an urgent public health issue. Antibiotic resistance of bacteria is a challenge that modern medicine has to face, it is increasingly said that we are approaching the beginning of the post-antibiotic era. According to the World Health Organization, recently approved antibiotics and those currently in clinical trials are not sufficient to spread drug resistance.
REFERENCES (43)
1.
Khardori N, Stevaux C, Ripley K. Antibiotics: From the Beginning to the Future: Part 2. Indian J Pediatr. 2019;87(1):43–47. https://doi.org/10.1007/s12098....
2.
Hutchings MI, Truman AW, Wilkinson B. Antibiotics: past, present and future. Curr Opin Microbiol. 2019;51:72–80. https://doi.org/10.1016/j.mib.....
3.
Ghosh D, Veeraraghavan B, Elangovan R, et al. Antibiotic Resistance and Epigenetics: More to It than Meets the Eye. Antimicrob Agents Chemother. 2020;64(2):e02225–19. 10.1128/AAC.02225-19.
4.
Gogineni V, Chen X, Hanna G, et al. Role of symbiosis in the discovery of novel antibiotics. J Antibiot (Tokyo). 2020;73(8):490–503. https://doi.org/10.1038/s41429....
5.
Hadjadj L, Baron SA, Diene SM, et al. How to discover new antibiotic resistance genes? Expert Rev Mol Diagn. 2019;19(4):349–362. https://doi.org/10.1080/147371....
6.
Talapko J, Meštrović T, Juzbašić M, et al. Antimicrobial Peptides-Mechanisms of Action, Antimicrobial Effects and Clinical Applications. Antibiotics (Basel). 2022;11(10):1417. https://doi.org/10.3390/antibi....
7.
Miethke M, Pieroni M, Weber T, et al. Towards the sustainable discovery and development of new antibiotics. Nat Rev Chem. 2021;5(10):726–749. https://doi.org/10.1038/s41570....
8.
Lesiuk M, Paduszyńska M, Greber KE. Synthetic Antimicrobial Immunomodulatory Peptides: Ongoing Studies and Clinical Trials. Antibiotics (Basel). 2022;11(8):1062. https://doi.org/10.3390/antibi....
9.
Uddin TM, Chakraborty AJ, Khusro A, et al. Antibiotic resistance in microbes: History, mechanisms, therapeutic strategies and future prospects. J Infect Public Health. 2021;14(12):1750–1766. https://doi.org/10.1016/j.jiph....
10.
Markiewicz Z, Korsak D, Popowska M. Antybiotyki w dobie narastającej lekooporności. Warszawa: Wydawnictwo Naukowe PWN; 2021.
11.
Terreni M, Taccani M, Pregnolato M. New Antibiotics for Multidrug-Resistant Bacterial Strains: Latest Research Developments and Future Perspectives. Molecules. 2021;26(9):2671. https://doi.org/10.3390/molecu....
12.
Zhang C, Straight PD. Antibiotic discovery through microbial interactions. Curr Opin Microbiol. 2019;51:64–71. https://doi.org/10.1016/j.mib.....
13.
World Health Organization, 2021 Antibacterial agents in clinical and preclinical development: an overview and analysis. Geneva, 2022.
14.
Sato T, Yamawaki K. Cefiderocol: Discovery, Chemistry, and In Vivo Profiles of a Novel Siderophore Cephalosporin. Clin Infect Dis. 2019;69(Suppl 7):S538–S543. https://doi.org/10.1093/cid/ci....
15.
De Oliveira DMP, Forde BM, Kidd TJ, et al. Antimicrobial Resistance in ESKAPE Pathogens. Clin Microbiol Rev. 2020;33(3):e00181–19. https://doi.org/10.1128/CMR.00....
16.
Kocsis B, Gulyás D, Szabó D. Delafloxacin, Finafloxacin, and Zabofloxacin: Novel Fluoroquinolones in the Antibiotic Pipeline. Antibiotics (Basel). 2021;10(12):1506. https://doi.org/10.3390/antibi....
17.
Mancuso G, Midiri A, Gerace E, et al. Bacterial Antibiotic Resistance: The Most Critical Pathogens. Pathogens. 2021;10(10):1310. https://doi.org/10.3390/pathog....
18.
Antibacterial products in clinical development for priority pathogens. https://www.who.int/observator... (access: 2023.03.06).
19.
Iskandar K, Murugaiyan J, Hammoudi Halat D, et al. Antibiotic Discovery and Resistance: The Chase and the Race. Antibiotics (Basel). 2022;11(2):182. https://doi.org/10.3390/antibi....
20.
Huemer M, Mairpady Shambat S, Brugger SD, et al. Antibiotic resistance and persistence-Implications for human health and treatment perspectives. EMBO Rep. 2020;21(12):e51034–e51034. https://doi.org/10.15252/embr.....
21.
Miller MJ, Liu R. Design and Syntheses of New Antibiotics Inspired by Nature›s Quest for Iron in an Oxidative Climate. Acc Chem Res. 2021;54(7):1646–1661. https://doi.org/10.1021/acs.ac....
22.
Minamimura M, Taniyama Y, Inoue E, et al. In vitro antibacterial activity and beta-lactamase stability of CP-70,429 a new penem antibiotic. Antimicrob Agents Chemother. 1993;37(7):1547–51. https://doi.org/10.1128/aac.37....
23.
Al-Tawfiq JA, Momattin H, Al-Ali AY, et al. Antibiotics in the pipeline: a literature review (2017–2020). Infection. 2022;50(3):553–564. https://doi.org/10.1007/s15010....
24.
Dunne MW, Aronin SI, Das AF, et al. Sulopenem for the Treatment of Complicated Urinary Tract Infections Including Pyelonephritis: A Phase 3, Randomized Trial. Clin Infect Dis. 2023;76(1):78–88. https://doi.org/10.1093/cid/ci....
25.
Shapiro AB, Moussa SH, McLeod SM, et al. Durlobactam, a New Diazabicyclooctane β-Lactamase Inhibitor for the Treatment of Acinetobacter Infections in Combination With Sulbactam. Front Microbiol. 2021;12:709974–709974. https://doi.org/10.3389/fmicb.....
27.
Hernández-García M, García-Castillo M, Ruiz-Garbajosa P, et al. In Vitro Activity of Cefepime-Taniborbactam against Carbapenemase-Producing Enterobacterales and Pseudomonas aeruginosa Isolates Recovered in Spain. Antimicrob Agents Chemother. 2022;66(3):e0216121–e0216121. https://doi.org/10.1128/aac.02....
28.
Hamrick JC, Docquier JD, Uehara T, et al. VNRX-5133 (Taniborbactam), a Broad-Spectrum Inhibitor of Serine- and Metallo-β-Lactamases, Restores Activity of Cefepime in Enterobacterales and Pseudomonas aeruginosa. Antimicrob Agents Chemother. 2020;64(3). https://doi.org/10.1128/aac.01....
29.
Dowell JA, Dickerson D, Henkel T. Safety and Pharmacokinetics in Human Volunteers of Taniborbactam (VNRX-5133), a Novel Intravenous β-Lactamase Inhibitor. Antimicrob Agents Chemother. 2021;65(11):e0105321. https://doi.org/10.1128/aac.01....
30.
Asempa TE, Kuti JL, Nascimento JC, et al. Bronchopulmonary disposition of IV cefepime/taniborbactam (2–0.5 g) administered over 2 h in healthy adult subjects. J Antimicrob Chemother. 2023;78(3):703–709. https://doi.org/10.1093/jac/dk....
31.
Papp-Wallace KM, Bethel CR, Caillon J, et al. Beyond Piperacillin-Tazobactam: Cefepime and AAI101 as a Potent β-Lactam-β-Lactamase Inhibitor Combination. Antimicrob Agents Chemother. 2019;63(5):e00105–19. https://doi.org/10.1128/AAC.00....
32.
Kaye KS, Belley A, Barth P, et al. Effect of Cefepime/Enmetazobactam vs Piperacillin/Tazobactam on Clinical Cure and Microbiological Eradication in Patients With Complicated Urinary Tract Infection or Acute Pyelonephritis: A Randomized Clinical Trial. JAMA. 2022;328(13):1304–1314. https://doi.org/10.1001/jama.2....
33.
Butler MS, Paterson DL. Antibiotics in the clinical pipeline in October 2019. J Antibiot (Tokyo). 2020;73(6):329–364. https://doi.org/10.1038/s41429....
34.
Bradford PA, Miller AA, O’Donnell J, et al. Zoliflodacin: An Oral Spiropyrimidinetrione Antibiotic for the Treatment of Neisseria gonorrheae, Including Multi-Drug-Resistant Isolates. ACS Infect Dis. 2020;6(6):1332–1345. https://doi.org/10.1021/acsinf....
35.
Scangarella-Oman NE, Hossain M, Hoover JL, et al. Dose Selection for Phase III Clinical Evaluation of Gepotidacin (GSK2140944) in the Treatment of Uncomplicated Urinary Tract Infections. Antimicrob Agents Chemother. 2022;66(3):e0149221–e0149221. https://doi.org/10.1128/AAC.01....
36.
Barrera CM, Mykietiuk A, Metev H, et al. Efficacy and safety of oral solithromycin versus oral moxifloxacin for treatment of community-acquired bacterial pneumonia: a global, double-blind, multicentre, randomised, active-controlled, non-inferiority trial (SOLITAIRE-ORAL). Lancet Infect Dis. 2016;16(4):421–30. https://doi.org/10.1016/s1473-....
37.
File TM, Rewerska B, Vucinic-Mihailovic V, et al. SOLITAIRE-IV: A Randomized, Double-Blind, Multicenter Study Comparing the Efficacy and Safety of Intravenous-to-Oral Solithromycin to Intravenous-to-Oral Moxifloxacin for Treatment of Community-Acquired Bacterial Pneumonia. Clin Infect Dis. 2016;63(8):1007–1016. https://doi.org/10.1093/cid/ci....
38.
Chen MY, McNulty A, Avery A, et al. Solithromycin versus ceftriaxone plus azithromycin for the treatment of uncomplicated genital gonorrhoea (SOLITAIRE-U): a randomised phase 3 non-inferiority trial. Lancet Infect Dis. 2019;19(8):833–842. https://doi.org/10.1016/s1473-....
39.
Flamm RK, Rhomberg PR, Sader HS. In Vitro Activity of the Novel Lactone Ketolide Nafithromycin (WCK 4873) against Contemporary Clinical Bacteria from a Global Surveillance Program. Antimicrob Agents Chemother. 2017;61(12):e01230–17. https://doi.org/10.1128/AAC.01....
40.
Cho JC, Crotty MP, Pardo J. Ridinilazole: a novel antimicrobial for Clostridium difficile infection. Ann Gastroenterol. 2019;32(2):134–140. https://doi.org/10.20524/aog.2....
41.
Tamma PD, Aitken SL, Bonomo RA, et al. Infectious Diseases Society of America Guidance on the Treatment of Extended-Spectrum β-lactamase Producing Enterobacterales (ESBL-E), Carbapenem-Resistant Enterobacterales (CRE), and Pseudomonas aeruginosa with Difficult-to-Treat Resistance (DTR P. aeruginosa). Clin Infect Dis. 2020;72(7):e169–e183. https://doi.org/10.1093/cid/ci....
42.
Yahav D, Shepshelovich D, Tau N. Cost Analysis of New Antibiotics to Treat Multidrug-Resistant Bacterial Infections: Mind the Gap. Infect Dis Ther. 2021;10(1):621–630. https://doi.org/10.1007/s40121....
43.
Tacconelli E, Carrara E, Savoldi A, et al. Discovery, research, and development of new antibiotics: the WHO priority list of antibiotic-resistant bacteria and tuberculosis. Lancet Infect Dis. 2018;18(3):318–327. https://doi.org/10.1016/S1473-....
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.